Free Courses Sale ends Soon, Get It Now


Free Courses Sale ends Soon, Get It Now


Disclaimer: Copyright infringement not intended.
Context
Highlights of the MoU
Significance
Benefits of International recognition of the Indian pharmacopoeia! Standards
[Note: The Indian Pharmacopoeia (IP) is officially recognized by five (5) countries: Afghanistan, Ghana, Nepal, Mauritius and the Republic of Suriname. The Ministry seeks to expand the nations which recognize the IP.]
|
INDIAN PHARMACOPOEIA COMMISSION (IPC) Indian Pharmacopoeia Commission (IPC) is an Autonomous Institution of the Ministry of Health and Family Welfare, Govt. of India. IPC is created to set standards of drugs in the country. Its basic function is to update regularly the standards of drugs commonly required for treatment of diseases prevailing in this region. It publishes official documents for improving Quality of Medicines by way of adding new and updating existing monographs in the form of Indian Pharmacopoeia (IP). It further promotes rational use of generic medicines by publishing National Formulary of India. IP prescribes standards for identity, purity and strength of drugs essentially required from health care perspective of human beings and animals. IPC also provides IP Reference Substances (IPRS) which act as a finger print for identification of an article under test and its purity as prescribed in IP. INDIAN PHARMACOPOEIA (IP) Indian Pharmacopoeia (IP) is published by the Indian Pharmacopoeia Commission (IPC) on behalf of the Ministry of Health & Family Welfare, Government of India in fulfillment of the requirements of the Drugs and Cosmetics Act, 1940 and Rules 1945 thereunder. IP is recognized as the official book of standards for the drugs being manufactured and/or marketed in India. IP contains a collection of authoritative procedures of analysis and specifications of drugs for their identity, purity and strength. The standards of the IP are authoritative in nature and are enforced by the regulatory authorities for ensuring the quality of drugs in India. During quality assurance and at the time of dispute in the court of law the IP standards are legally acceptable. The Standards prescribed in the IP are to establish compliance with regulatory requirements on an article. The criteria to be adhered to are: The interpretation of a monograph must be in accordance with all the general requirements, testing methods, texts, and notices pertaining to it, in the IP. A product is not of standard quality unless it complies with all the requirements of the monograph. |
India Suriname Relations in detail:
https://www.iasgyan.in/daily-current-affairs/india-suriname-relations
https://www.iasgyan.in/daily-current-affairs/india-suriname-relations-12
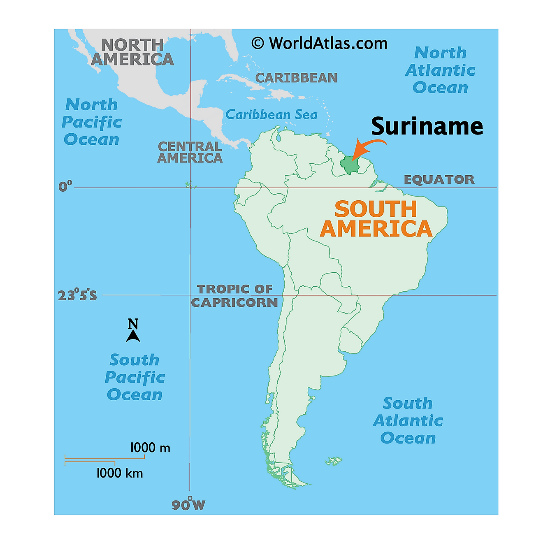
Suriname
Location
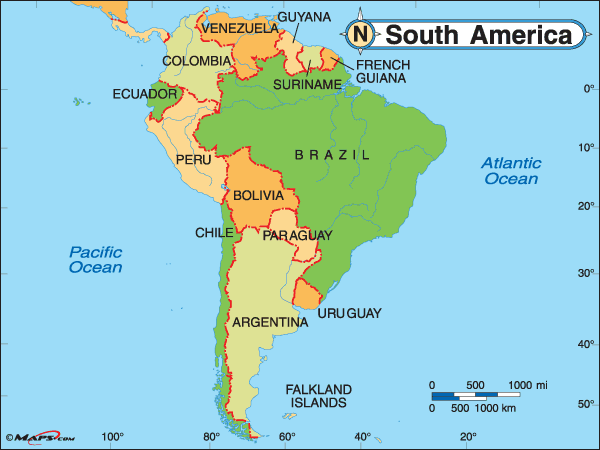
Borders
Geographic Regions
Mountain Ranges
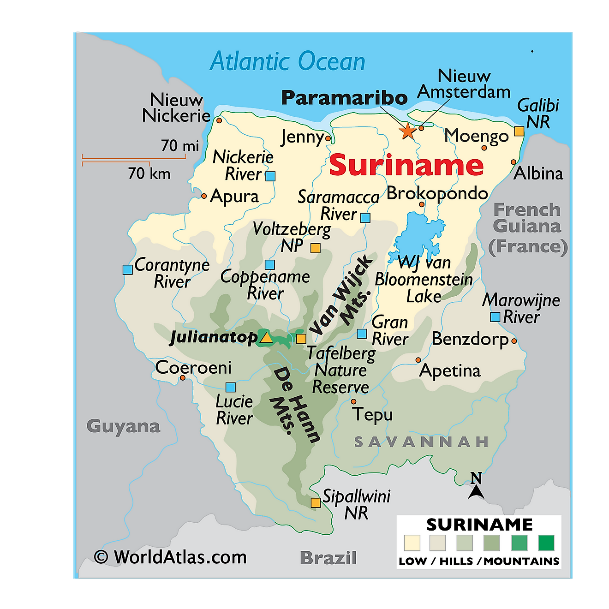
Ecoregions
Climate
Biodiversity and Conservation
|
PRACTICE QUESTION Q. Consider the following statements: 1. Suriname is the smallest independent country in African Continent situated on the Guiana Shield. 2. Julianatop is the highest mountain in the country. 3. Its forest cover is 90.2%, the highest of any nation in the world. How many of the above are correctly matched? A) Only 1 B) Only 2 C) All 3 D) None Answer: B) Only 2 |
https://pib.gov.in/PressReleasePage.aspx?PRID=1949414
© 2024 iasgyan. All right reserved